Wow. It has been 60 days since George’s bone marrow transplant. So far, we have just been chugging along. George is working full time from his home office, getting his magnesium transfusions every day, home infusion team comes twice a week to draw labs, I’m making sure he eats even though he doesn’t want to. All in all, things have been going smoothly.
But on August 23, we got a call from Rite-Aid saying George’s medicine was ready to be picked up. Huh? We didn’t call in any refills….and he wasn’t prescribed anything new as far as we knew. So we thought maybe it was an old prescription and the call was just a mistake. Later in the day, George got a notification that he had a message from the team at UPenn. When he opened it, it read:
Your most recent CMV level was 910. We would like to start a different anti-viral medication to help treat your most recent cmv reactivation. I have sent the the new medication to your rite aid pharmacy. The new medication is valgancyclovir 900 mg twice daily. Once you start the new medication please stop the acyclovir.
We will continue to recheck your CMV level once weekly. Once your level is negative again we will stop the valgancylovir and restart acyclovir.
The most common side effect of the valgancylovir is that it can affect your blood counts.
Wait, what? What is CMV?? (consults Google). So, Cytomegalovirus (CMV) is a well-known pathogen in immunosuppressed patients. After primary infection, this betaherpesvirus is not eradicated but establishes life-long infection in its host. CMV is dispersed and becomes dormant in multiple end organs, a state also referred to as “latency,” and can later be reactivated by a number of different stimuli, including immunosuppression and inflammation. Approximately 60% of people in the United States have been infected with CMV by the age of 6 years, and thus a large population harbor latent virus, making them “at risk” for reactivation.
Basically, like all viruses I the herpes family, once you get this you have it forever. So, it was lying dormant in George and once his immune system was suppressed, it was free to “reactivate.”
He was tested for CMV before the transplant and he did have it in his system. We just didn’t pay attention to those test results because we didn’t know what the implications were.
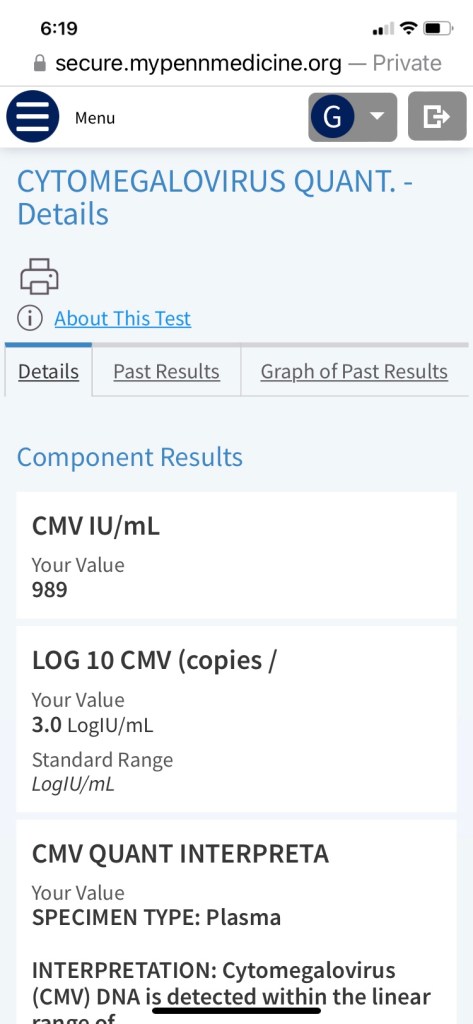
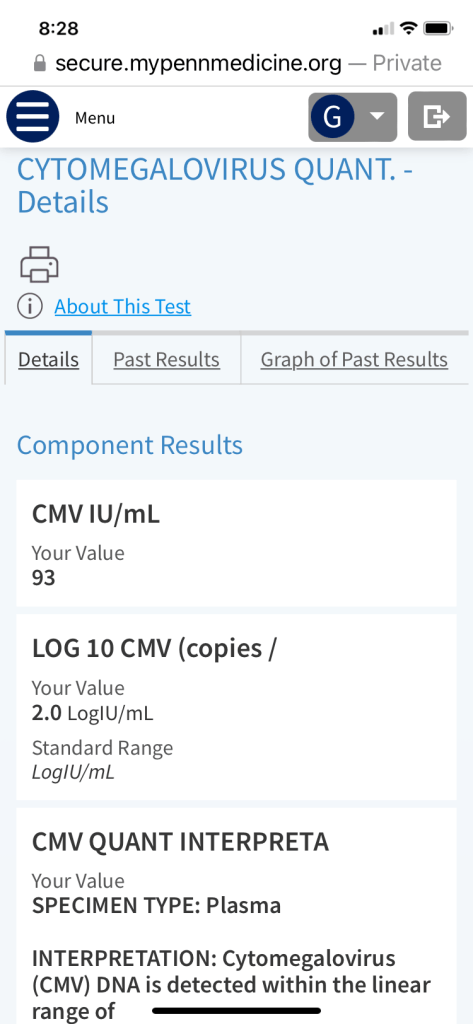
We were glad to see his count coming down nicely after a week of taking the new anti-viral medication.
When we first got the news about the CMV reactivation, we of course immediately consulted Google, as I previously stated. Pro-tip: don’t do that. Talk to your doctor! Because, what did we find? Well, this: For people who have weakened immune systems, especially people who have had an organ, stem cell or bone marrow transplant, CMV infection can be fatal. Oh, ok! Reading that and all the things that could go wrong with CMV reactivation was pretty scary and distressing. But, it seems like the risks are great for untreated CMV, and George is being treated. So, it was a relief to see those numbers come down and to read the message he received again and realize, there was no sense of urgency or panic in it and the plan is to return him to his regularly scheduled anti-viral once the CMV is under control. This is one of the reasons he gets his blood drawn twice a week. To monitor for things like this.
George met with Dr. Loren for the first time since June on August 31. It was super encouraging for George because she told him he looks amazing and she was astounded to learn he was working every day and that he is able to walk over 2 miles. She said his blood counts are great, that everything looks great! Exactly what we wanted to hear!

George’s next doctor’s appointment is on September 13. I noticed something in Dr. Loren’s notes after George’s appointment on Aug 31 that I want to ask about. She wrote:
“consider post-transplant gilteritinib given low level FLT3 diagnosis.”
Gilteritinib, sold under the brand name Xospata, is an anti-cancer drug. It acts as an inhibitor of FLT3. It was developed by Astellas Pharma and in April 2018, Astellas filed a new drug application with the Food and Drug Administration for gilteritinib for the treatment of adult patients with FLT3 mutation–positive relapsed or refractory acute myeloid leukemia (AML).
The FLT3 gene contains instructions for a protein called FLT3, which helps white blood cells grow. Yes, George was found have the FLT3 mutation when he had his first bone marrow biopsy. And yes, that moved him from a good prognosis to fair/poor. That along with the ASXL1 mutation. But….why is the doctor considering this treatment when it’s for relapsed or refractory (not achieving remission) AML? George hasn’t even had a bone marrow biopsy after his transplant yet to see if there is any residual disease- he will have one next month. So, this is confusing and somewhat alarming. Is she anticipating refractory AML? We will definitely be asking Jackie, the bone marrow transplant guru, on our next visit.
Also, months ago we asked Dr. Loren if the fair/poor prognosis due to the genetic mutations pertained to before transplant and if that prognosis would change once he had new bone marrow and was in remission. We never really got an answer. I am going to ask that again.

Leave a comment